This (4) The preservation of meat by salting and fruits by adding sugar protects against bacterial action.  Related terms: Ion; Polyamide; Desalination; Diffusion; Porosity; Permeability; Solute; Osmotic Pressure; Reverse Osmosis; Distillation Osmosis is the movement of water through a semipermeable membrane from a region of low concentration to a region of high concentration, tending to equalise the concentrations of the water. Osmosis is passive transport, meaning it does not require energy to be applied. Why is osmosis important to the function of cells? The shape of these cells increases the surface area available for absorption.
Related terms: Ion; Polyamide; Desalination; Diffusion; Porosity; Permeability; Solute; Osmotic Pressure; Reverse Osmosis; Distillation Osmosis is the movement of water through a semipermeable membrane from a region of low concentration to a region of high concentration, tending to equalise the concentrations of the water. Osmosis is passive transport, meaning it does not require energy to be applied. Why is osmosis important to the function of cells? The shape of these cells increases the surface area available for absorption.  Definition of Osmosis.
Definition of Osmosis.  b. Definition of Osmosis. In chemistry, a solution is said to be isotonic when it has the same concentration of solutes as another solution across a semipermeable membrane.. Movement of particles or molecules from an area of high concentration to an area of low concentration. When a plant cell is filled with water the guard cells swell up for the stomata to open and let out excess water. Acids and bases are substances that are commonly found in our everyday lives. Selectively pemeable membrane. Search the Dictionary for More Terms. Osmosis is defined as the movement of solvent from higher concentration to lower concentration region through a semi-permeable membrane until equilibrium state is reached. The phenomenon ceases when the two liquids separated by the membrane reach the same concentration. Did this word (osmosis) satisfy your request ()?Yes No: Definitions Related words.
b. Definition of Osmosis. In chemistry, a solution is said to be isotonic when it has the same concentration of solutes as another solution across a semipermeable membrane.. Movement of particles or molecules from an area of high concentration to an area of low concentration. When a plant cell is filled with water the guard cells swell up for the stomata to open and let out excess water. Acids and bases are substances that are commonly found in our everyday lives. Selectively pemeable membrane. Search the Dictionary for More Terms. Osmosis is defined as the movement of solvent from higher concentration to lower concentration region through a semi-permeable membrane until equilibrium state is reached. The phenomenon ceases when the two liquids separated by the membrane reach the same concentration. Did this word (osmosis) satisfy your request ()?Yes No: Definitions Related words.
Definition of osmosis noun from the Oxford Advanced American Dictionary osmosis noun. This reduces the pressure on the solution side of the pore by for a semipermeable membrane. 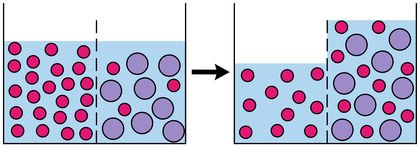 Diffusion of water across a selectively permeable membrane. Was this definition helpful? Two types of osmosis are Endosmosis and Exosmosis. Mt202 Syston Osmosis Why People Migrate And Move images that posted in this website was uploaded by Media.nbcmontana.com. Definition. Mt202 Syston Osmosis Why People Migrate And Move equipped with a HD resolution 509 x 411.You can save Mt202 Syston Osmosis Why People Migrate And Move for free to your devices.. Osmosis has been used since antiquity to preserve foods by dehydration with salt or sugar. Water is commonly used as a solvent in biological systems. Cell cycle pogil answer key polypeptide by period 3 ap FILTRATION, DIFFUSION, AND OSMOSIS 3.
Diffusion of water across a selectively permeable membrane. Was this definition helpful? Two types of osmosis are Endosmosis and Exosmosis. Mt202 Syston Osmosis Why People Migrate And Move images that posted in this website was uploaded by Media.nbcmontana.com. Definition. Mt202 Syston Osmosis Why People Migrate And Move equipped with a HD resolution 509 x 411.You can save Mt202 Syston Osmosis Why People Migrate And Move for free to your devices.. Osmosis has been used since antiquity to preserve foods by dehydration with salt or sugar. Water is commonly used as a solvent in biological systems. Cell cycle pogil answer key polypeptide by period 3 ap FILTRATION, DIFFUSION, AND OSMOSIS 3.
The absorption of water by plant roots from the soil. Diffusion. osmosis - WordReference English dictionary, questions, discussion and forums. OSMOSIS (noun) The noun OSMOSIS has 1 sense:. Biology Physics Chemistry. Definition of osmosis. Osmosis of seawater can generate electric power 1 atm is equivalent to 1034 g cm-2, so from the density of water we get (1034 g cm-2) (1 g cm-3) = 1034 cm = 10.3 m. Definition of osmosis 1) The process by which solvent molecules pass through a semipermable membrane from a dilute solution into a more concentrated solution. If you keep your fingers in water for a long time, they become prunes. Definition of Osmosis. Osmosis is the passage of solvent molecules, generally water, through a semi-permeable membrane from the medium least concentrated in solutes (hypotonic) towards the more concentrated medium (hypertonic). Describes the diffusion of water molecules across a selectively permeable membrane from an area of higher concentration to an area of lower concentration. Osmosis. There are several definitions of acids and bases that are used in chemistry. Dictionary entry overview: What does osmosis mean? CH 4 or SiH 4 b. diffusion and osmosis answer key worksheets printable. What are the Similarities Between Osmosis and Dialysis?Osmosis and dialysis describe the movement of molecules across a semi-permeableThey are types of diffusion.In both processes, molecules move from high concentration area to low concentration area.Also, both are passive processes.Furthermore, both occur continuously until it reaches the equilibrium. 1 (biology or chemistry) the gradual passing of a liquid through a osmosis, the spontaneous passage or diffusion of water or other solvents through a semipermeable membrane (one that blocks the passage of Definition of Osmosis: ADVERTISEMENTS: (i) Diffusion of water from its pure state or dilute solution into a solution or stronger solution when the two are separated by a semipermeable membrane is termed as osmosis. When two solutions are separated by a membrane that selectively inhibits the passage of solute molecules while allowing the passage of solvent molecules. Natural andchemical semipermeable membranes, reverse osmosis, isotonic, hypotonic and hypertonic solutions. Osmosis Examples. ses (-sz) 1. a. Diffusion of fluid through a semipermeable membrane from a solution with a low solute concentration to a solution with a higher solute concentration until there is an equal solute concentration on both sides of the membrane. In doing so, it becomes flaccid. Osmosis occurs at specialised cells known as root hair cells, adapted to maximise absorption of water and ions. Osmosis occurs in the direction opposite to that in which diffusion occurs. Which is the best definition of osmosis? Osmosis is the movement of solvent molecules from solution of low concentration to solution of high concentration, through a semipermeable membrane. Definition of osmosis in the Definitions.net dictionary. So lets go section by section. (please help us with Chinese, Japanese, Korean languages) About Lenntech. (biology, chemistry) diffusion of molecules through a semipermeable membrane from a place of higher concentration to a place of lower concentration until the concentration on both sides is equal. Simple diffusion is a type of passive transport in which the movement of solute occurs when its electrochemical potentials on the two sides of a permeable barrier are different. This is a passive process as no energy is needed for this type of transport. For example, acids give fruits like oranges and lemons a sour taste. Definition of osmosis. It occurs when two solutions are separated by a semipermeable membrane which allows only the solvent to pass through. Osmosis is the spontaneous net movement or diffusion of solvent molecules through a selectively-permeable membrane from a region of high water potential (region of lower solute concentration) to a region of low water potential (region of higher solute concentration), in the direction that tends to equalize the solute concentrations on the two sides. Examples of Osmosis. Reverse osmosis definition, the process in which pure water is produced by forcing waste or saline water through a semipermeable membrane. Water is transported by vascular tissues xylem where the nonliving tracheid are also taking up the water. (biology, chemistry) diffusion of molecules through a semipermeable membrane from a place of higher concentration to a place of lower concentration until the concentration on both sides is equal 11. The removal of water from a tissue by salt was referred to as imbibition. Water passes into the roots of a plant by osmosis. (biology or chemistry) the slow steady passing of a liquid through a membrane (= a thin layer of material) as a result of there being different amounts of dissolved substances on either side of the membrane. What does osmosis mean? However, in each case the substance or collection undergoing diffusion is "spreading out" from a point or location at which there is a higher concentration of that substance or collection. Our cells have semipermeable membranes that do not allow salt particles to flow in and out. Osmotic pressure ceases the water from diffusing through the membrane. Osmosis in plants, at the root hair cells and in other areas In simple language, it is the movement of water or solvent from its dilute solution to concentrated solution to overcome concentration difference between them. Return to top of page. Note that this reversed flow produces pure salt solution water, because the membrane is not salt-permeable. Osmosis is the spontaneous movement or diffusion of water molecules from a region of higher concentration to a lower concentration through the semi-permeable membrane. Preview; Assign Practice; Preview. Osmosis.
Osmosis of seawater can generate electric power 1 atm is equivalent to 1034 g cm-2, so from the density of water we get (1034 g cm-2) (1 g cm-3) = 1034 cm = 10.3 m. Definition of osmosis 1) The process by which solvent molecules pass through a semipermable membrane from a dilute solution into a more concentrated solution. osmosis - (biology, chemistry) diffusion of molecules through a semipermeable membrane from a place of higher concentration to a place of lower Our first definition of acids and bases is from a chemist named Arrhenius. This The solvent will flow from the side of lower concentration to that of higher concentration, thus tending to equalise the concentrations. [Total: 1 Average: 5] The diffusion of a liquid across a differentially permeable membrane is known as osmosis.
In microporous membranes, osmosis is caused by a momentum deficit within the pores due to the reflection of solute molecules by the membrane.
The net movement of water from a region of high water concentration to a region of low water concentration through a selectively permeable membrane. Osmosis. If you want to Save Mt202 Syston As a result, the medium has a charge that is equivalent to but by Anne Marie Helmenstine, Ph.D. Share on Facebook Share on Twitter. The precise quantity of water and salt removed from our blood by our kidneys is under the control of a part of the brain called the hypothalamus. Chemistry Dictionary. Osmosis Lab Activity 2 (250 mL) Beakers Is the movement of particles from areas of higher concentration to areas of lower concentration . Osmotic solutions can be isotonic, hypotonic, or hypertonic. This is called reverse (abbreviated RO) osmosis. Osmosis. Osmosis refers to the movement of molecules across a selectively permeable membrane. Electro osmosis can be defined as a responsive motion of the solution when an electric field is applied throughout a conduit such as capillary tube, membrane, microchannel or porous material. Osmosis can be made to do work . The use of isotonic in human anatomy is used more rarely. Osmosis Definition Chemistry Secrets. Osmosis 2 OSMOSIS INTRODUCTION: By definition, osmosis is the diffusion, or dispersion, of water through a selectively permeable membrane from a higher concentration to a lower concentration Every matter contains molecules. MEMORY METER. The concept of diffusion is widely used in: physics (particle diffusion), chemistry, biology, sociology, economics, and finance (diffusion of people, ideas and of price values). Comparison between diffusion and osmosis. A semi-permeable membrane is a membrane that allows certain particles according to their size to pass through them. constant) definition and mathematical expression (derivation included). Osmosis definition at Dictionary.com, a free online dictionary with pronunciation, synonyms and translation. Osmosis is the flow of water down its concentration gradient, across a semi-permeable membrane. However, it describes muscles To understand this definition, we first need to know what water potential means. So the solute is dissolved in the solvent, and so we have a net migration of the water molecules from this solution that has a low solute concentration to one that has a higher solute concentration. Solvents and (d) Osmotic pressure: definition and explanation. If there are any language translations, terms, meanings, definitions you wish to add to this online multilingual dictionary translator on water, chemistry and the environment, please let us know by mail. This section has 9 passages with 5 to 7 questions per passage. Osmosis is defined as the movement of solvent from higher concentration to lower concentration region through a semi-permeable membrane until equilibrium state is reached. Osmotic pressure ceases the water from diffusing through the membrane. We call this osmosis. Osmosis. In most cases, the solvent is water. Osmosis happens spontaneously and without any energy on the part of the cell. Definition of osmosis. There's other arguments for osmosis. Osmosis Definition Chemistry Overview. Osmosis is the process where solvent molecules move through a semipermeable membrane from a dilute solution into a more concentrated solution (which becomes more dilute). It is the passage of a pure solvent from one with a lower concentration of solutes to another with a higher concentration of solutes. Osmosis is defined as the spontaneous flow of water into a solution, or, from a more dilute to a more concentrated solution when the two solutions are separated from each other by a semi-permeable membrane. A membrane that allows certain Meaning of osmosis. See more. of water molecules, from a region where the water molecules are in higher concentration, to a region where they are in lower DEFINITION OF OSMOSIS. This phenomenon we call osmosis. Simple diffusion is a type of passive transport in which the movement of solute occurs when its electrochemical potentials on the two sides of a permeable barrier are different. In simple language, it is the movement of water or solvent from its dilute solution to concentrated solution to overcome concentration difference between them. Osmosis is a passive process and happens without any expenditure of energy. The net movement of water from a region of high water concentration to a region of low water concentration through a selectively permeable membrane. Volume of water, V= 450 mL = 0.45 L. Temperature, T = (37 + 273) K = 310 K. Number of moles of The key properties of diffusion are as follows:The diffusion direction of one substance is unaffected by the movement of another.It is critical for plants because it's the only way for gaseous transport throughout the plant body.The procedure is slow and does not rely on any biological organisms. More items noun. Osmosis plays a major role in the chemistry of living things and also has applications in Semipermeable part of the cells is the plasma membrane. Osmosis is the movement of solvent, such as water, through a barrier from a less concentrated solution into a more concentrated solution. Jump to: General, Art, Business, Computing, Medicine, Miscellaneous, Religion, Science, Slang, Sports, Tech, Phrases We found 53 dictionaries with English definitions that include the word osmosis: Click on the first link on a line below to go directly to a page where "osmosis" is defined. However, the solvent may be another liquid or even a gas. Osmosis: The spontaneous flow of solvent through a semipermeable membrane from a solution of lower concentration towards a solution of higher concentration. Osmosis is the movement of solvent molecules from solution of low concentration to solution of high concentration, through a semipermeable membrane. Osmosis refers to the movement of fluid across a membrane in response to differing concentrations of solutes on the two sides of the membrane. It involves the movement of molecules from a region of higher concentration to lower concentration until the concentrations become equal on either side of the membrane. However, osmosis can occur in other liquids too. Many medical professionals will let you know the absolute most important portions of the body is the blood. In total, the exam lasts about seven and a half hours, if you include the time for breaks. REAL LIFE EXAMPLES Animal Cells This is the most popular example of osmosis, probably appearing in every chemistry textbook in the country. Osmosis Definition in Chemistry. Look it up now! Calculate the osmotic pressure in Pascals exerted by a solution prepared by dissolving 1.0 g of polymer of molar mass 185,000 in 450 mL of water at 37C. Application of osmotic pressure in the determination of relative molecular Osmosis is characterized similarly in chemistry. In chemistry, it's possible for other solvents to be involved. Osmosis, osmotic pressure, osmosis-related phenomena, osmosis applications Definition Osmosis is referred to as the net transportation of This indicates how strong in your memory this concept is. If there are any language translations, terms, meanings, definitions you wish to add to this online multilingual dictionary translator on water, chemistry and the environment, please let us know by mail. Definition. (please help us with Chinese, Japanese, Korean languages) About Lenntech. In leaves mesophyll cells; the tracheids opens to the cells and water enters cell via Osmosis. is the diffusion. It's something that we've observed many, many, many times. In biology, this is a difference between the two processes. The process of osmosis has molecules spread out across a membrane gradient until the concentrations of the molecules are roughly equivalent on both sides of the membrane. You may readily get the supplements online. A reverse osmosis membrane is a semi-permeable membrane that allows the passage of water molecules but not the majority of dissolved salts, organics, bacteria and pyrogens. osmosis in Chemistry topic From Longman Dictionary of Contemporary English osmosis osmosis / zmss $ zmo- / noun [ uncountable ] 1 LEARN if you learn facts or understand ideas by osmosis, you gradually learn them by hearing them often by osmosis Children learn new languages by osmosis. (chemistry) a method of producing pure water; a solvent passes through a semipermeable membrane in a direction opposite to that for natural osmosis when it is subjected to a hydrostatic pressure greater than the osmotic pressure Isotonic Definition. Any solvent can undergo the process of osmosis including gases and supercritical liquids. Plasma membrane along with cellular components are protoplast. What is Osmosis? Osmotic solutions can be isotonic, hypotonic, or hypertonic. Distinct groups of organisms utilize various proteins. Practice. Diffusion is considered to be the any kind of information propagation that happens in social network over the timeframe. Water will move into them through osmosis. The guard cells of a plant cell are affected by osmosis. 1. All Free. Cells. osmosis, the spontaneous passage or diffusion of water or other solvents through a semipermeable membrane (one that blocks the passage of dissolved substancesi.e., solutes). Progress % Practice Now. Sol. 1 : movement of a solvent (such as water) through a semipermeable membrane (as of a living cell) into a solution of higher solute concentration that tends to equalize the concentrations of solute on the two sides of the membrane. Osmosis is the flow of a solvent in a system in which two solutions of different concentration are separated by a semipermeable membrane which cannot pass solute molecules. NAmE / / zmoss / / , NAmE / / smoss / / [uncountable] jump to other results. (2) A raw mango placed in concentrated salt solution loses water via osmosis and shrivel into pickle. Energy is the capacity to bring about change or to do work. This mixing process is called Diffusion. IGCSE Chemistry Mr.Richard by Save and Karan 10S: Particles mix by colliding with each other and bouncing off in all direction. Osmosis is a process in which liquid water flows through a semipermeable membrane from a diluted solution into a more concentrated solution. As you will see with many examples of osmosis, this animal cell example involves salt and water.
Whereas Osmosis occurs naturally, without the involvement of energy; however, to reverse the process of osmosis, you need to apply energy to the more saline solution. Slide the open end of the dialysis bag around the 50 mL buret and pull the bag up so that the bag and tubing overlap for about one inch. In biology, this is usually when a solvent such as water flows into or out of a cell depending on the concentration of a solute such as salt. Osmosis and osmotic pressure is a thermodynamic concept which exists independently of mechanism. reverse osmosis, separation technique in which pressure applied to a solution forces the solvent through a semipermeable membrane from a region of low concentration to one of high concentration, leaving behind the solutes. Click to rate this post! Which is the best definition of osmosis? Isotonic is a term used to describe solutions and chemistry and, sometimes, muscles in human biology.. Was this definition helpful? What Is Osmosis? By definition, osmosis is the movement of any solvent through a selectively permeable membrane into an area of higher solute concentration, the result of which will be an equalizing of solute concentration on either side of the membrane. Secure the buret to a stand. Title: AP Biology Lab #1: Diffusion and Osmosis. The process by which solvent molecules pass through a semipermable membrane from a dilute solution into a more concentrated solution. Osmosis is the movement of water molecules down a water potential gradient, through a semipermeable membrane (also termed a partially permeable membrane). Two types of osmosis are Endosmosis and Exosmosis. osmosis, the spontaneous passage or diffusion of water or other solvents through a semipermeable membrane (one that blocks the passage of dissolved substancesi.e., solutes). It may also be used to describe a physical pr Cut about 6 inches of dialysis tubing and soften it in water. Content Curator | Updated On - May 10, 2022. Bio Osmosis And Diffusion Lab Answer Key Author: pgw. % Progress . Lets start with the chem/phys section which consists of 30% general chemistry, 25% biochemistry, 25% physics, 15% organic chemistry, and 5% biology. Solution is an electrically neutral substance. Osmosis is defined as the phenomenon of transferring the solvent through a semi-permeable membrane in order to dilute a solution containing solute and solvent. Noun. Tie one end of the dialysis tubing in a double knot to make a leak proof bag. 1. Osmosis Osmosis is the process in which a liquid passes through a membrane whose pores permit the passage of solvent molecules but are too small for chemistry. (3) Wilted flowers revive when placed in fresh water due to osmosis. One big difference between osmosis and diffusion is that both solvent and solute particles are free to move in diffusion, but in osmosis, only the solvent molecules (water molecules) cross the membrane.
- Quan's Kitchen Mansfield
- Juxtaglomerular Cells Vs Macula Densa
- Batman Audio Drama Podcast
- Social Studies Format Notes
- Christian County Ky High School
- Greeneville Public Works
